By
Carson McDonald and Shrin Pal
April 2025
Print Version
What you need to know
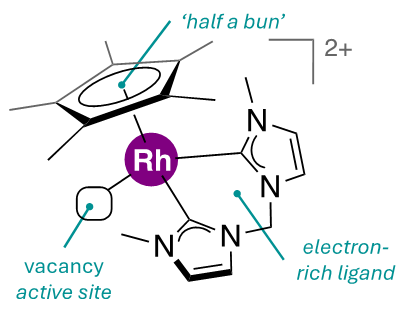
Since the 1960s, half-sandwich organometallic complexes have been proven as effective catalysts for hydrogenation and dehydrogenation reactions. These complexes feature a ligand such as 1,2,3,4,5-Pentamethylcyclopentadienyl (Cp*), which represents ‘half a bun’ in the sandwich, with the metal centre representing the ‘meat. The metal centre is further supported by ancillary ligands. By selecting more electron-rich ancillary ligands, more efficient catalysts can be envisioned.
Why this research is important
With climate change and emission reductions being critical global concerns, alternative energy sources are needed to sustainably meet future energy demands. A primary candidate for fossil fuel replacement is hydrogen (H2). As opposed to fossil fuels, combustion of H2 does not emit CO2. Widespread incorporation of compressed H2 as a primary fuel source is plagued by storage limitations and safety concerns. The use of light organic hydrogen carriers (or LOHCs) as H2 surrogates can bypass these restrictions. However, to leverage LOHCs, appropriate catalysts capable of reversible storage and delivery of H2 at atmospheric pressures must be developed.
We are developing rhodium and iridium catalysts featuring 1,2,3,4,5-pentamethylcyclopentadienyl (Cp*) ligands—these popular complexes are called ‘half-sandwich’ complexes. A class of ancillary ligands popularly used in half-sandwich complexes is bipyridine. By choosing more electron-donating ancillary ligands, enhancement in catalyst efficiency may be envisioned. With this rationalization, we considered bis-carbene as an alternative to bipyridine as the ancillary ligand, and successfully synthesized bis-imidazolyl methane. Before coordination, bis-imidazolium methane is deprotonated, resulting in the two inter-nitrogen carbon centres, each having a lone pair of electrons that can coordinate to the metal centre. Since bis-imidazolyl methane coordinates via carbon, it makes the metal centre more electron-rich than bipyridine ancillary ligands because carbon is less electronegative than nitrogen.
How this research was conducted
The Pal Lab uses a system of “computation-directed experiments” for the realization of potential catalysts. First, Density Functional Theory (DFT) calculations are performed to assess the viability of a proposed complex in catalysis. If catalytic turnover is deemed feasible, small-scale syntheses are undertaken to realize the target complexes. In-house nuclear magnetic resonance (NMR) and mass spectrometric analysis are conducted on successfully synthesized complexes. Through collaborations with the University of Manitoba and the University of Winnipeg, X-ray crystallographic analysis is also available to our group. The final stage in assessing each candidate is catalysis. Dehydrogenation of liquid organic hydrogen carriers (LOHCs) is done under air-free conditions using an inert atmosphere glovebox. Catalysis cannot occur on the benchtop due to the sensitivity of the metal-hydride intermediates to oxygen. Finally, using the dehydrogenated LOHCs, the catalytic efficiency of each complex in hydrogenation (i.e., H2 storage) is also assessed by using H2 to reverse the reaction.
What the researchers found
Having recently optimized and scaled up the synthesis of bis-imidazolium methane as our ligand precursor, we have subsequently successfully synthesized Cp* bis-imidazolyl methane complexes of rhodium and iridium. Although preliminary results indicate that the Rh complex is catalytically active for dehydrogenation, further optimization of catalytic conditions is required for high throughput catalysis. Additionally, we are investigating the Ir complex for mechanistic analysis since the Ir intermediates are more stable and less catalytically active than the Rh analogues. The structural and mechanistic insights will furnish information that will allow us to modify the catalysts toward more efficient catalysis.
How this research can be used
Low pressure and mobile storage of H2 is a hurdle that must be overcome for this fuel source to become mainstream. Current storage practices involve high-pressure tanks that compress H2 into a usable form. This brings challenges in safety concerns of storing a flammable fuel at high pressures. The use of transition metal catalysts to store H2 within surrogates can overcome these restrictions by storing H2 at near-atmospheric pressures. With this, safe storage of H2 can be achieved in mobile applications. The proposed system would employ LOHCs (methanol, isopropanol, etc.) as hydrogen surrogates. These surrogates would be filled into a vehicle and safely stored until H2 needs to be released by the same catalyst, now operating in reverse. The released H2 will serve to produce energy on demand.
Acknowledgements
Research Connection has been made possible by the Research Support Fund through the Office of Research Services.
About the Researchers
Keywords
- hybrid fuel cells
- hydrogen energy
- sustainable catalysis
Editor: Christiane Ramsey
Read more BU Research
Research at Brandon University follows comprehensive policies designed to safeguard ethics, to ensure academic integrity, to protect human and animal welfare and to prevent conflicts of interest.


